What Are The Types of Clinical Trials?

Every year, thousands of clinical trials take place across the United States. Here at the United Research Medical Institute (UMRI), we conduct many high-quality clinical trials at our facility.
You may have heard what clinical trials are actually for. Well, when a research study is done with human volunteers, it is known as a clinical trial.
These trials only take place when there is a likelihood that patients could get better care from a new treatment or test. In fact, doctors have to prove that there is a chance the new treatment may be better than existing options.
Volunteering for a Clinical Trial
If you ever want to participate as a volunteer for a clinical trial, you can choose to join any of the different phases. In order to decide which one is best for you, consider how your health is, and determine the risk levels of each stage.
By taking part in a trial, you can make a significant contribution to the advancement of science. Volunteers with UMRI are able to receive access to new treatments for ophthalmology conditions before they become available for prescription use. These participants also receive diagnostic testing, hematologic testing, and physician care.
Furthermore, clinical trial volunteers typically get compensated for their time and for following instructions according to the protocol. Each clinical trial has a different protocol with diverse exclusion and inclusion criteria to determine who is able to participate in the study.
Preclinical Trials
To prove their assertion, doctors will do preclinical research to assess the factors involved in the potential new test or treatment. This work is not done on human volunteers, but rather on laboratory animals to minimize potential harm.
Preclinical research on new treatments can test different things such as potential effects on the mind, impacts on the body, and chemical composition.
Once this preclinical research is completed, it is time to start the various stages of clinical trials. These stages will serve to test whether the potential treatment is safe for human use and effective as a solution. There are five different phases of clinical trials with each occurring in a specific order to keep participants safe.
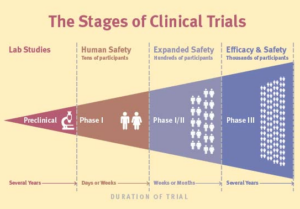
Source: brainandlife.org Infographic: Anthony Wing Kosner
Phase 1 of a Clinical Trial
During phase 1 of a clinical trial, clinicians aim to find the best version of a test or treatment. Ideally, they want to find the one with the fewest side effects and highest levels of safety.
Doctors may collect information about:
- The test or treatment
- How and when volunteers underwent the test or treatment
- Potential issues or side effects
- The impact of the test on volunteers
Doctors begin this process by providing small doses of the treatment to a handful of volunteers. Larger doses are given to other participants until side effects become too severe or the desired effect is noted.
The overall goal of Phase 1 trials is to get an idea of how safe a drug may be. If after Phase 1, the treatment is deemed safe, it is allowed to be tested in Phase 2 of the clinical trial.
UMRI Phase 1 First in Human (FIH) Trials
Here at UMRI, we are the only Phase 1-4 ophthalmology unit in the United States. We can hold patients overnight and create a controlled environment which is key to improving the quality of our clinical trials.
Over the last twenty years, we have conducted over 130 studies relating to:
- Dry eye syndrome
- Glaucoma
- Diabetic eye diseases
- Infusions
- Optical devices
- Healthy normal subject trials
- And more
Our board-certified ophthalmologist, Dr. James Peace, has years of expertise in Phase 1-4, first in human (FIH), early-phase research. We also specialize in Phase 1 FIH studies in particular.
These trials are very important for researchers looking to understand the effects of a new treatment or test on human beings. FIH trials are able to shed light on how safe a treatment may be for the human population and how it starts to work to address the problem at hand.
Conclusion
In conclusion, clinical trials are very important for testing the effectiveness and safety of potential new treatments and tests. Volunteers play a key role in enabling researchers to understand how these treatments affect the human body.
Here at UMRI, we pride ourselves on advancing research that helps improve lives. By conducting studies in phases 1 – 4 and specializing in first in human trials, we are able to test innovative solutions that hold great promise to the medical world.
To volunteer or learn more about UMRI and our studies, please click here.